Nuclear Stability is a concept that helps to identify the stability of an isotope. The excess neutrons act somewhat like nuclear glue. These extra neutrons are necessary for stability of the heavier nuclei. It can be observed from the chart that there are more neutrons than protons in nuclides with Z greater than about 20 (Calcium). Atoms such as 1H, 2H whose nuclei contain the same number of protons but different number of neutrons (different A) are known as isotopes.Ītomic Number and Nuclear Stability Segre chart – This chart shows a plot of the known nuclides as a function of their atomic and neutron numbers. 2H is the hydrogen nuclide with a neutron as well as a proton in the nucleus (2H is also called deuterium or heavy hydrogen). Thus the symbol 1H refers to the nuclide of hydrogen with a single proton as nucleus. Each nuclide is denoted by chemical symbol of the element (this specifies Z) with tha atomic mass number as supescript. 
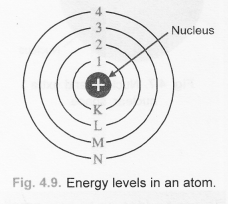
The various species of atoms whose nuclei contain particular numbers of protons and neutrons are called nuclides. The total number of nucleons, that is, protons and neutrons in a nucleus, is equal to Z + N = A, where A is called the mass number. The number of neutrons in a nucleus is known as the neutron number and is given the symbol N. Hydrogen (H), for example, consist of one electron and one proton. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. Since the number of electrons is responsible for the chemical bavavior of atoms, the atomic number identifies the various chemical elements. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19coulombs. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
